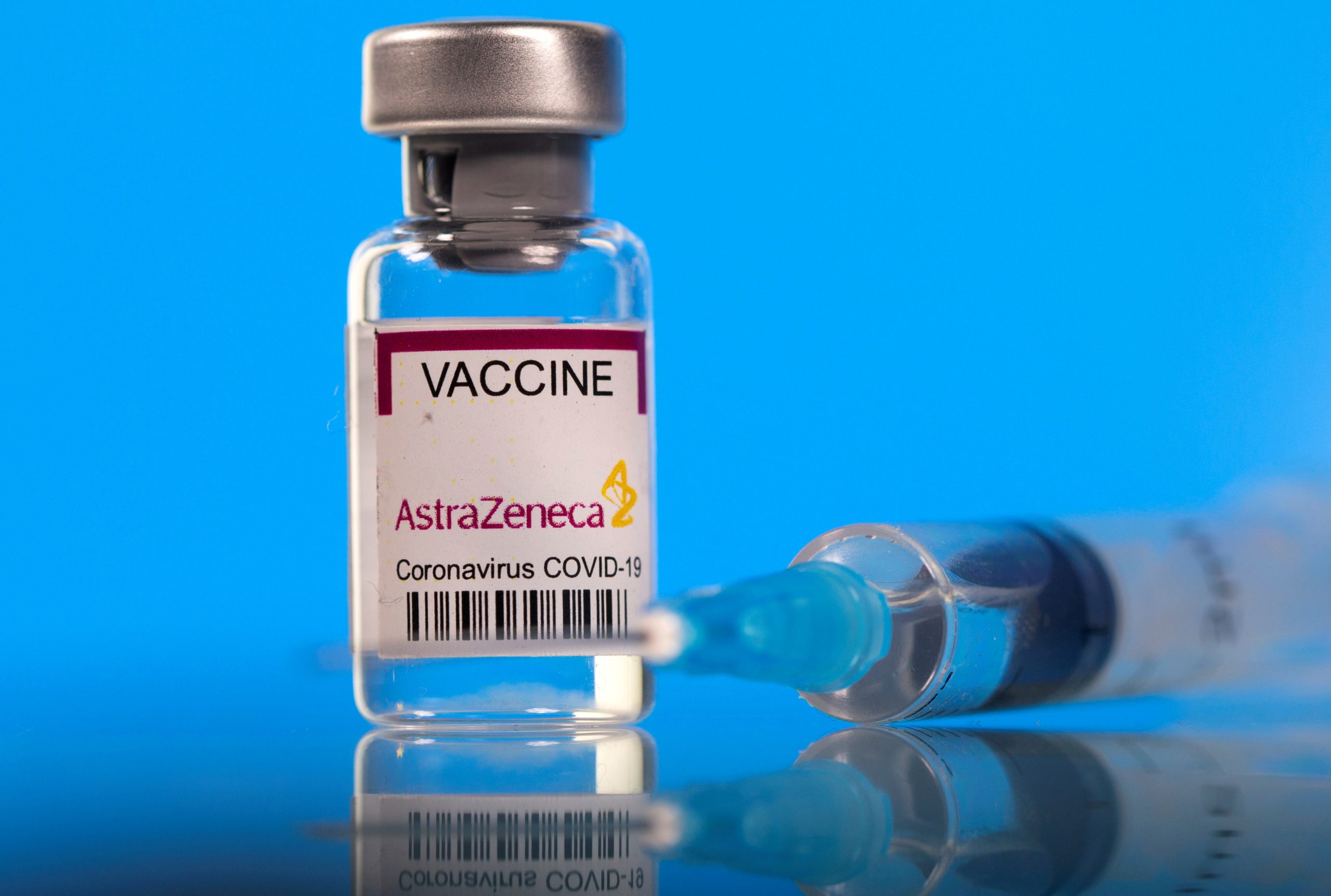
AstraZeneca’s said its COVID-19 vaccine was 76% effective in a new analysis of its U.S. trial – only a tad lower than the level in an earlier report this week criticised for using outdated data.
Interim data published on Monday had put the vaccine’s efficacy rate at 79% but had not included more recent infections, leading to a highly unusual public rebuke from U.S health officials.
The small revision to the efficacy rate will go a long way to putting the vaccine back on track for gaining U.S. emergency use authorisation – which it plans to seek in the coming weeks – and help AstraZeneca in its efforts to dispel doubts about its effectiveness and side-effects, independent experts said.
AstraZeneca also reiterated that the shot, developed with Oxford University, was 100% effective against severe or critical forms of the disease. There have been eight severe cases – all among trial participants who received the placebo.
“The vaccine efficacy against severe disease, including death, puts the AZ vaccine in the same ballpark as the other vaccines,” said William Schaffner, an infectious disease expert from the Vanderbilt University School of Medicine, adding that he expects the shot to gain U.S. approval.
The latest data was based on 190 infections among more than 32,400 participants in the United States, Chile and Peru. The earlier interim data was based on 141 infections through Feb. 17.
It also said the vaccine showed 85% efficacy in adults 65 years and older, higher than the 80% rate reported on Monday.
AstraZeneca said the latest data has been presented to the independent trial oversight committee, the Data Safety Monitoring Board, and it plans to submit the analysis for peer-reviewed publication in the coming weeks.
“The primary analysis is consistent with our previously released interim analysis, and confirms that our COVID-19 vaccine is highly effective in adults,” Mene Pangalos, executive vice president of BioPharmaceuticals R&D at AstraZeneca said in a statement.
The drugmaker noted there were 14 additional possible or probable cases to be analysed so numbers in later updates of the trial results may fluctuate slightly.
The updated 76% efficacy rate compares with rates of about 95% for vaccines from Pfizer/BioNTech and Moderna in their trial data.
Experts have noted, however, that AstraZeneca’s latest data is particularly significant because it was compiled after more infectious variants of the coronavirus became prevalent.
“This appears to be a very effective vaccine with no safety concerns,” said Paul Griffin, a professor at the University of Queensland.
“Hopefully, this should now give people the confidence that this vaccine is the right one to continue to use moving forward,” he said, adding that he and his parents have received the vaccine.
AstraZeneca’s vaccine is seen as crucial in tackling the spread of COVID-19 across the globe, not just due to limited supply of vaccines but also because it is easier and cheaper to transport than rival shots. It has been granted conditional marketing or emergency use authorisation in more than 70 countries.
The vaccine, once hailed as a milestone in the fight against the COVID-19 pandemic, has been dogged by questions since late last year when the drugmaker and Oxford University published data from an earlier trial with two different efficacy readings as a result of a dosing error.
Then this month, more than a dozen countries temporarily suspended giving out the vaccine after reports linked it to a rare blood clotting disorder in a very small number of people.
The European Union’s drug regulator said last week the vaccine was clearly safe, but Europeans remain sceptical about its safety.
Canada on Wednesday said it was safe but added a warning to the vaccine’s label about rare blood clots.
Its rollout has also been marred by production glitches and export curbs imposed by India and the European Union, threatening to slow global efforts to end the pandemic which has killed more than 2.8 million.
(Reporting by Rocky Swift in Tokyo, Swati Pandey in Sydney, Peter Henderson in San Francisco and Shubham Kalia in Bengaluru; Writing by Miyoung Kim; Editing by Edwina Gibbs)




















 Continue with Google
Continue with Google